
We'll probably have to wait for 2016 to see the next official version of ISO 13485. If it were approved, a final draft (FDIS) would be released in 2015 and the new official version in 2016. there will be a new round of comments / changes / approval, which makes the objective of publishing the final version in 2015 very unlikely.Ī new draft (DIS) may be released by the end of the year or the beginning of next year.Result of last WG1 meetingĭuring the last WG1 meeting held in july, the published draft version of ISO 13485 (the one called ISO DIS 13485:201X, you've probably heard about or had a copy of the draft) was submitted to the vote of the WG members. You can pay a visit to the TC 210 page on ISO website here and browse the list of standards managed by this committee. WG1 is the Working Group in charge of writing standards about the Application of quality systems to medical devices, namely ISO 13485.ISO TC 210 is the Technical Committee of the ISO organization, in charge of Quality management and corresponding general aspects for medical devices,.Finally, the organization must be re-certified every three years in order to maintain its ISO 13485 certification status.The working group in charge of writing the next version of ISO 13485 is the: ISO TC 210 WG1.(See Who is able to grant certification ) If you pass this audit, the Registrar issues an ISO 13485 Certificate demonstrating that your organization is Registered to ISO 13485 for a three-year period. Then a Certification Body (CB or Registrar) audits the performance of your organization against the latest version of the ISO 13485 Requirements.Follow the steps to implement an ISO 13485 quality management system.To become ISO 13485 certified, your organization must: This then allows them to audit other companies. An individual, however, CAN become an ISO 13485 Certified Lead Auditor after a 5-day training course. Instead, an organization or company becomes certified. It is NOT a personal standard – a person cannot get certified to ISO 13485.This is a process-based standard: you use it to control your processes, then your end product should meet the desired results. It does NOT matter what size your organization is: 1 person or 1 million people.In the meantime, the medical device industry can rely on AAMI TIR102:2019, which is a bi-directional mapping tool that was released on August 30, 2019. FDA 21 CFR 820 with ISO 13485:2016, making ISO 13485 the FDA’s mandatory QMS for Medical Devices (the rule is expected to be released in 2019). the FDA has proposed a rule which would harmonize U.S. This global standard is mandatory in some countries, and in the U.S. In addition, the documentation and safety requirements are much greater under ISO 13485:2016, whereas ISO 9001:2015 puts focus on customer satisfaction and continuous improvement. (Yes, 2008, not 2015.) This is because it does not follow the high-level structure (Annex L) of the latest version of ISO 9001 (which is 9001:2015). While ISO 13485:2016 remains a stand-alone document, it aligns with ISO 9001:2008.
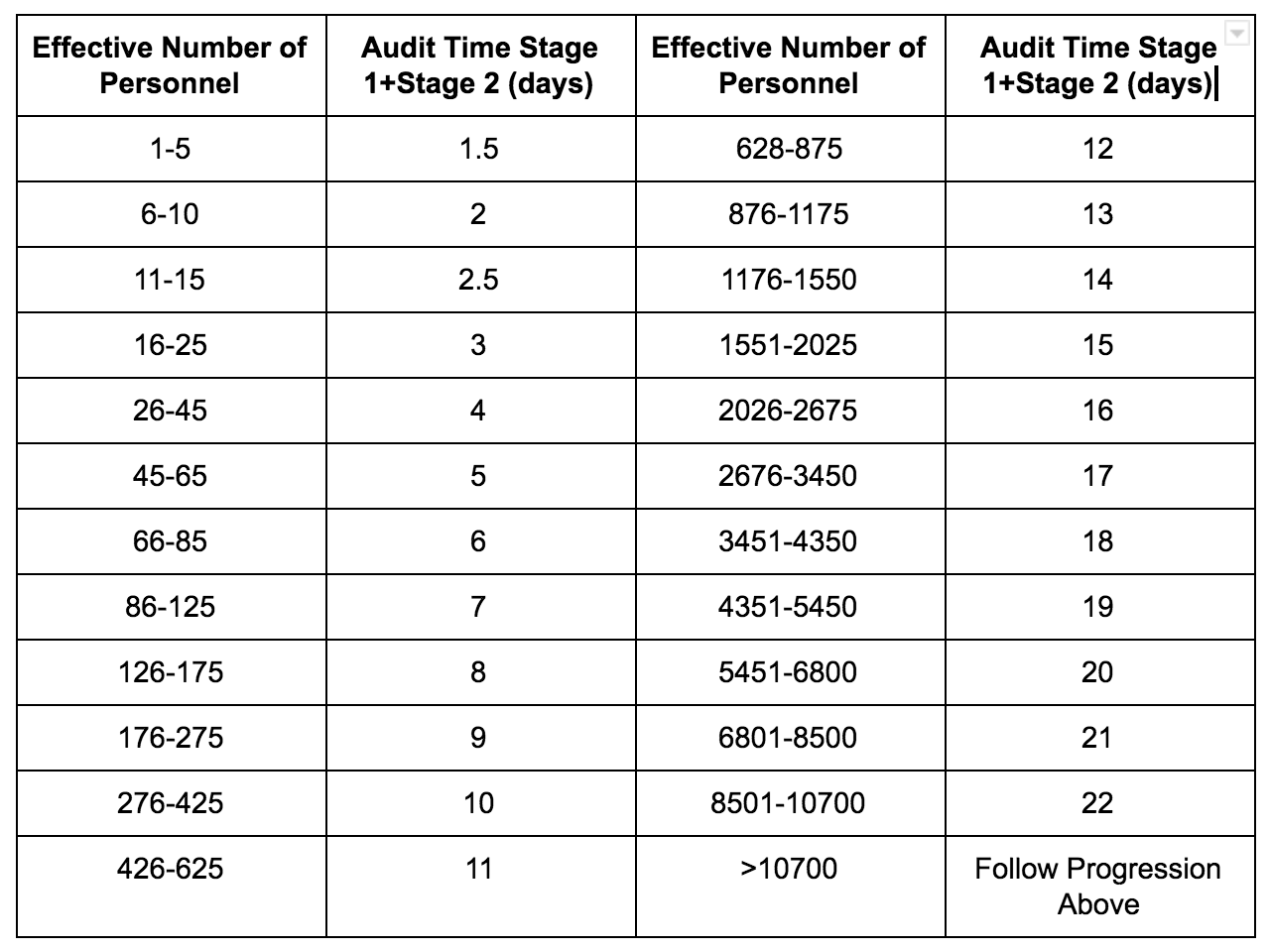
For the most part: ISO 13485 = ISO 9001 + Additional Medical Device Requirements. ISO 13485:2016 is the standard for a Quality Management System (“QMS”) for the design and manufacture of Medical Devices.Ĭertification to the standard requires an organization’s quality management system to pass a third-party Medical Device Single Audit Program, or “MDSAP” Audit. ISO 13485 PowerPoint Training Materials.ISO 13485 Online Training Course Listing.ISO 13485 Internal Auditor Training Courses.ISO 13485 Lead Auditor Training Courses.Simple Steps to ISO 13485 Certification Follow our proven and manageable step-by-step process for a successful ISO implementation project.
